Chapter:
Interphase-Mass-Transfer
1. Diffusion of components between phases at equilibrium is
2. The operating line will be straight if its concentrations are represented in mole ratio.
3. Consider a steady-state condition; concentration at any point in equipment never changes with time.
4. Find operating line slope
Where, X,Y- concentration in mole ratio of liquid and gas phase
x,y-concentration in mole fraction
Gs,Ls- mole free basis( non-diffusing solvent)
5. According to Lewis and Whitman ory, departure from concentration equilibrium at Interphase is due to
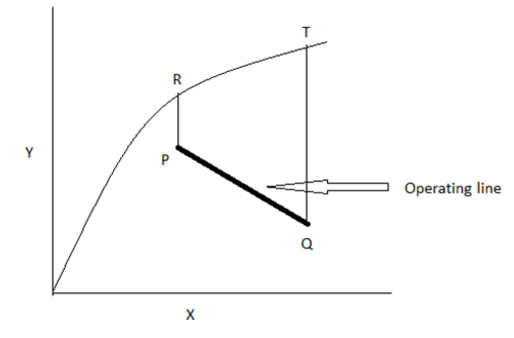
6. The figure given below is a single stage ideal co- current process of solute transfer from liquid to gas phases.
7. Name of line PR and QT
If X, Y – are represented in mole ratio
8. The real driving force of mass transfer is
9. Assume an ideal solution with pure component A, has a vapour pressure of 300 mmHg and total pressure of 1atm. The concentration in terms of mole ratio is
10. Find abscissa and ordinates of graph
If X,Y –Concentration in mole ratio
x,y- Concentration in mole fraction
All Chapters
View all Chapter and number of question available From each chapter from Mass-Transfer
Modes and Diffusion
Modes and Diffusion
Interphase Mass Transfer
Interphase Mass Transfer
Equipment for Gas-Liquid Operations
Equipment for Gas-Liquid Operations
Humidification Operations
Humidification Operations
Gas Absorption
Gas Absorption
Distillation
Distillation
Liquid Extraction
Liquid Extraction
Drying
Drying
Leaching and Mass Transfer Co-efficients
Leaching and Mass Transfer Co-efficients
Diffusion in Solids and Molecular Diffusion in Fluids
Diffusion in Solids and Molecular Diffusion in Fluids
Topics
This Chapter Interphase-Mass-Transfer consists of the following topics
 Guest
Guest